|
In this lab, we learned about atoms and how they bond in a inorganic compound and organic molecules. We learned about Lewis dot structures and how to assemble the electrons when we draw them. We learned that atoms either give electrons or gain electrons in order to make elements. When the atoms bond with other atoms, they have to find a way to get 8 valence electrons (or 2 valence electrons for hydrogen), this is called the octet rule. We practiced making bonds by drawing Lewis dot structures on a worksheet. We had to draw out at least 10 molecules to show our understanding of the dot structures. Then, once we were done with that, used the 10 bonds we did to make physical models of the bonds using gum drops, raisins, and tooth picks. The gum drops represented different elements, the raisins represented hydrogen and the tooth picks represented bonds. After we constructed physical bonds for the inorganic compounds, we had to construct 3 organic molecules.
What I noticed about the shapes of the molecules is that in the inorganic compounds is that most of them have a central base of where the other elements are attached too. For organic molecules, I noticed that they usually form a hexagon and have some bonds attached to the hexagon. I feel like drawing the Lewis dot structures helped us see where the atoms bond with the other atoms, we saw where we should place the certain atoms when we make the physical models of the atoms. In this lab, I have learned about the smell of molecules. Also I have learned that similar looking molecules tend to look the same. For example, in our lab we smelled a family of molecules called alcohols and phenols and two molecules stood out to me that looked similar. Those two molecules/items were oregano and thyme. I've noticed that their molecule structures looked similar and having just a little difference of where the oxygen and hydrogen molecules were placed in both structures. And other than that, they are both herbs that have a similar leafy look.
Also to support this claim is two molecules/items in the ketones family. These two are peppermint and spearmint. The structure of both molecules look similar because they both have a hexagon and three stick looking bonds sticking out of the hexagon. Both of them smell really similar because they are both mints. They both also have a similar looks because they are both leafy mints. Overall in this lab I have learned that many molecules that look similar, there is a big chance that the actual look of the molecule/item will look similar too. As I look through my blog, I realized that my reflections have gotten longer as the semester ended. My first few posts are very brief but still got the point across. I think at the beginning, I was very lost on what was expected to be a good reflection because there wasn't a sample of a great reflection so I just did what I could. But as the semester went along, Andrew went over what was expected of us many times and I believe that my reflections improved overtime by just adding little details and sentences I never usually brought up in my older reflections. Though the semester, I struggled getting the 250 because I never really knew how many points I got per post so when I added up my estimated points I believe I got but then when Andrew grades them, the two numbers would be different. I liked it when Andrew did blog checks, so I knew where I stood, whether I needed to add more to my blog or to start prepping for the next section. I know most people don't like writing up the reflections, but I felt that at times I enjoyed writing up what I had just did and learned. I like them because they gave me a chance to research more in depth information on what I did and not just what was required to put in the proposal. But with that said, sometimes I just pushed them aside because I'd rather be doing other stuff for class and all the reflections just started building up near the end and I hated when that happened.
By writing these blog posts and labs, I learned that I had procrastinated a whole lot and I didn't really push to work with different people. I had always procrastinated throughout this semester but I mostly felt it during this last section, because I knew that I had worksheets done so I didn't really make an effort to start new labs. What I did do this section was shaping rocks and I really enjoyed that even though it took away most of the time I had in class to work. Because it took so much of my class time, I didn't really make an effort to write up the reflections for my previous labs until the very last week in the section. A struggle I had with procrastinating was that since I wrote the reflections days or weeks after the actual experience, I didn't really have a clear memory of what had happened. I feel that I didn't really work with new people because I was more comfortable with working with the people I usually hang out with. I felt awkward asking other people to do labs with me because they might be busy with other labs or I think they'd be more comfortable with doing labs with their friends too. I think I was the most motivated was in section 1 and beginning of section 2 because that was when I did all the labs I really wanted to do and then for the rest of the sections I just ran out of ideas. Although, it seems that I didn't do worksheets until the last section, but really all the worksheets are from through out the sections and I saved them so I could use them as last minute points if I didn't have enough. I learned that I actually liked the lessons Andrew gave us to go with the worksheets because I actually got to understand the background of what is actually happening to molecules while I am doing the labs. Also, I think I am pretty used to practicing what I have learned on a worksheet or at least something that would let myself know that I understood the information that was given to me. My favorite labs were the bath bomb lab and the pop rocks lab because those two items really interested me and I wanted to learn how they were made and worked so I can make them myself at home. Although the pop rocks didn't work the way we wanted to, it was still a fun learning experience. Now I know that I probably shouldn't try making the pop rocks at home because in the lab I had just made a huge piece of sour laffy taffy. I was the most motivated to learn about these two topics so I can know how to improve the next time. I wasn't as motivated in section 3 because I just did labs that I had found on google that I thought would be cool but I really procrastinated to writing the reflections and actually learning the science behind them so I only knew a brief on what is going on. What happened though, was that at the beginning of spring break, I decided that I was going to finish the reflections for the labs I have done so far, and I spent hours and hours just learning more about what I have done over the past two weeks. And by writing the reflections for those labs, I learned a whole bunch. And I actually enjoyed learning the science behind it and I just needed some motivation to learn it. But for those labs, I was mainly motivated to get them done before the section ends instead of being motivated for the knowledge. From this class, I will takeaway the management skills I had to develop at the beginning of this semester in order to get an A in this class. I had to learn how to manage my time and keep track of my points for the class. Also I will takeaway the knowledge that I had to learn and teach to myself of how to make certain items. For example, I have made bath bombs, jewelry, rocket engines, and many more. I also tookaway that knowledge can be achieved by self discovery and not just by what the teachers will teach. Lots of what I have done for this class was designed to my liking and I just had to get what I want to do approved by Andrew. There was a upside and a downside to that because at times I really loved doing what ever I wanted because It was fun but then somedays, I wouldn't know what to do at all and I just needed a teacher to tell me what to do. From left to right, the rocks I have shaped are amethyst, rose quartz, and obsidian. I really enjoyed shaping the rocks so I can wear them as jewelry. I came across multiple struggles with shaping the rocks but I had a lot of help from people who knew how to use the lapidary machine to educate me and help me with shaping them. The amethyst I have shaped was my first piece of rock I shaped and it didn't really turn out the way I planned it to turn out. What I wanted it to be shaped into was similar to how my rose quartz piece turned out. It turned out the way it is because I wasn't good on choosing a piece that was big enough without a lot of cracks in the rock. It was turning out pretty well until I became impatient and asked Jiapsi to cut my rock smaller to speed up the shaping process. He had warned me that It could crack because of how fragile the rock is and because there was already a crack forming but I told him to do it anyways. As he was cutting the amethyst, it had cracked in half so I was left with a small piece of it. With a small piece, I just decided to shape it into a circle so I can at least still have a piece of amethyst to wear as jewelry. For my rose quartz, I was a little more careful on decided whether or not to cut the rock. But I was pretty impatient on shaping this rock too until we had a new wheel come in which was the 100 diamond wheel. The wheel was coarser so it grinded my rock faster than before. With both of the rocks, I had trouble on deciding when they were done with the brown, red and blue wheel so I kept bothering Jiapsi so he can check if I am done with each wheel. Faceting the rocks were pretty difficult, and this goes for my obsidian piece too, because when I shaped the pointy end, the angles didn't always match up with the straight edges. I tried my best in matching them up but the results are still crooked. For my obsidian piece, I decide to give myself a challenge and I faceted both sides. AmethystAmethyst is a purple variety of quartz. The purple color is caused because of the iron that is in it. If heated, amethyst may turn yellow-orange, yellow-brown, or dark brownish, and then resembles the quartz variety citrine. The hardness of it is similar to quartz. A lot of amethyst is produced in the state of Minas Gerais in Brazil and It is usually in large geodes of volcanic rocks there. Other places amethyst could be found is in Russia, South Korea, the United States, Uruguay, Maissau, Austria, India and many more. Up until the 18th century, amethyst was included in the cardinal, or valuable gems such as diamond, emerald, ruby, and sapphire. It has lost it's value because a extensive amount of it was discovered in Brazil. It's crystallization habit is forming a 6-sided prism ending in 6-sided pyramid but it doesn't always for this structure. Amethyst is the birthstone of February and it is said to have power, healing, and protection energies. Rose QuartzRose quartz, a silicon dioxide crystal, is one of the most common varieties of quartz. It's name comes from the color it has, which ranges from pale pink to a deep reddish-pink. It usually occurs in massive form, but it sometimes grows in clusters of small prismatic crystals. It varies in clarity from opaque to translucent to a foggy transparency. The color is usually considered as due to trace amounts of titanium, iron, or manganese. The places that contain large amounts of rose quartz are Brazil, Mexico, Uruguay, Russia, France, Namibia and Morocco. When it forms it typically is a 6-sided prism ending in 6-sided pyramid. Quartz (rose quartz is a type of quartz) is the second abundant mineral on the earth's crust, after the mineral feldspar. ObsidianObsidian is volcanic glass formed as an extrusive igneous rock. It is formed when the lava is quickly cooled so rapidly that atoms are unable to arrange themselves into a crystalline structure. The result is a volcanic glass with a smooth texture that breaks with a conchoidal fracture. Obsidian is hard and brittle so when it breaks, there is usually very sharp edges. Pure obsidian's color is usually dark but sometimes it has different colors depending on the presence of impurities. Magnesium and iron usually give obsidian a black to brown color. In some obsidian, there are small white spots which are crystals of cristolbalite. The volcanic glass might contain gas bubbles from the lava flow and these bubbles can produce different effects like a gold sheen on the glass. An iridescent, rainbow-like sheen is caused by inclusions of magnetite nanoparticles. It is found in many place all around the world. Obsidian was used to make knives, arrowheads, spear points, scrapers, and many other weapons and tools. Thin blades of obsidian are placed in surgical scalpels used for some of the most precise surgery because obsidian can produce a cutting edge that is thinner and sharper than the best surgical steel.
Proposal:Essential Question:How is soap made? Materials:
Ingredients:
Instructions:
Clean up: When you’re done making soap, always clean your equipment that has been exposed to lye. You can neutralize the lye with white vinegar, then wash the equipment well as you normally would. For the rest of it, let it sit for several days. Why? Because when you first make soap, it’s all fat and lye. You’ll be washing forever and you could burn your hands on the residual lye. If you wait, it becomes soap and all it takes to clean it is a soak in hot water. Sources: Reflection:Looking Back:
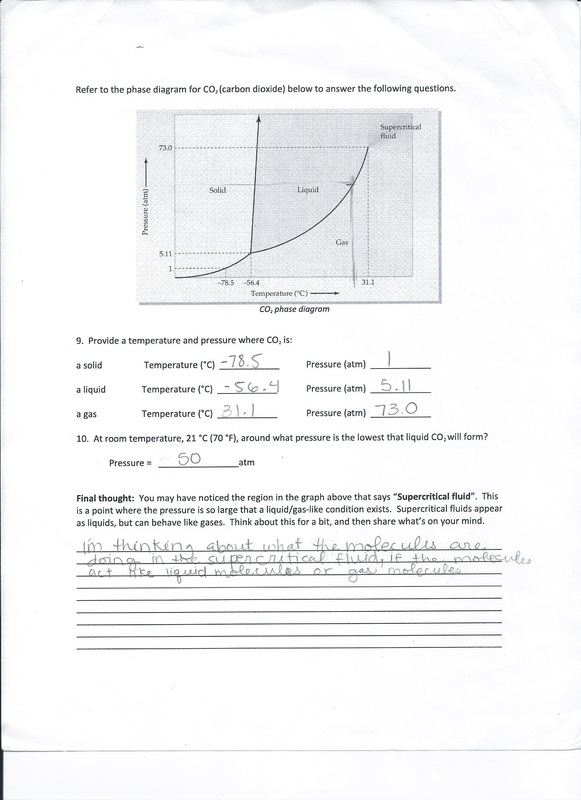
When we were done with mixing all the materials for the soap together, we got this goopy, pudding-like substance. at this point we had to move fast to put the substance in a mold or else it would harden and we cant mold it at all. My group didn't get to buy a mold because it would be too expensive to buy one if we were only going to make soap once. So we ended up using a small box and that ended up too big so we moved to using a plastic container lined in a plastic bag so it wouldn't stick to the container. It wasn't a great shape so our soap didn't turn out looking nice. In the slideshow above, you can see how it looked like, to me it looked like pancake mix. Looking Beyond: Saponification is the chemical reaction that happens when soap is being made. The reaction happens when oil and fats are mixed with a strong alkali. The strong alkali is usually lye which is also known as sodium hydroxide or potassium hydroxide. Oil has fatty acids which are carboxylic acids consisting of a long hydrocarbon chain at one end and a carboxyl group at the other end. Long chains of the fatty acids usually occur as triglycerides and are found in fats and oils. Triglycerides are esters of fatty acids and are formed by combining fatty acids with glycerol. When the fatty acids react with the glycerol, they form triesters of glycerol or triglyceride. During that process, the water molecules in the reaction is eliminated. When triglycerides in fat and oil react with the water molecules in the sodium hydroxide or potassium hydroxide, they are converted into soap and glycerin. Looking Inward: We used the soap and I think it worked out pretty well. The soap made our skin really soft and it felt as if it took off most of the oils on our skin. It also came out to be pretty bubbly which made it fun to use. One downside to me was that the smell of the soap wasn't really appealing, it smelt a lot like olive oil and not the lavender that we added. I think that since we decided to mix the oils in a immersion blender and if we didn't do that then it would have taken forever to get the oils to be at trace and without it being at trace then our soap wouldn't have turn out as good. I think I performed really good in this lab because I took charge of the lab and directed everyone what to do. I think I could have done a better job with getting my group to know what they are doing by making them help me with the proposal because then they'll actually have to educate themselves before writing the proposal. Having them do that would have helped me so much because I felt as if I was the only one knowing what was going on. Looking Outward: For this lab I worked with Daisy, Zion and Erika. I think we performed okay as a group. Most of the time I felt like I was doing most of the work because no one really helped me with the proposal. Also a couple days before we did the lab, I told my group to read the proposal thoroughly and when we did the lab, it showed that none of them even read the proposal beforehand. When we were doing the lab, they told me to slow down because they didn't know what was going on at the moment, but we were running low on time so I just had to keep on going to get the lab done. I was mostly the director of the lab tell everyone what to do and what to get next and watching over everything making sure everything is running smoothly. Looking Forward: If I were to do this lab again, I would for sure get soap molds!! Also, I would use the immersion blender right to mix everything instead of mixing it with a spoon because it would make the process more faster. The website I read told me that we could use a spoon if we want so we did that and 20 minutes pasted and we didn't see any progress in the soap so that was when we moved to use the blender and in under five minutes, we got the texture that we wanted. I wouldn't mind working with these people again but probably not all of them in the same group. I feel they all just assumed that I would do all the work and they'll be there for the lab and I didn't like that so I assume that if were with other people, the work would be distributed better. When researching about making soap, I was that there were many processes to make soap and different types of soaps. To continue the soap making lab, I would like to try to make glycerin soap, goat milk soap, the hot process of making soap and even laundry soap. Takeaway: In this lab I tookaway that if one method doesn't work, another method will for example, mixing with a spoon didn't work so we used the immersion blender. Also I learned that I need to be more better at distributing work evenly among my group so no one would be doing nothing or anyone is doing too much. I also learned that it is pretty easy to be cleaning while doing the lab, I saw that if someone wasn't doing anything while we were doing the lab I sent them to clean the materials that we were done using so we didn't have to clean much when we were done with the lab. |
AuthorMy name is Ivy and this is my tenth grade chemistry blog. Archives
November 2015
Categories |





 RSS Feed
RSS Feed
