What is the science happening when the ball is forming:
The bouncy ball that's is being made is made from a polymer. Polymers are molecules made up of similar chemical units bonded together. Glue contains polymer polyvinyl acetate (PVA) which is a type of polymer. The borax acts as a “cross-linker” to the polymer molecules in the glue. In other words, the borax helps the glue create chains of molecules that stay together when you pick them up. The cross link provides the ball with it's bouncing property. The chemical reaction between the glue and the borax is the main reaction that is going on in the lab. Also in the lab is corn starch, the corn starch also reacts with the borax and glue to help bind the molecules together so the shape would hold up better.
Proposal:
Essential Question: What makes bouncy balls bouncy?
Sources:
http://chemistry.about.com/od/demonstrationsexperiments/ss/bounceball.htm
http://www.hometrainingtools.com/a/make-a-colorful-bouncy-ball
http://www.pbs.org/parents/crafts-for-kids/super-bouncy-balls/
Materials:
Everything should be washable after the experiment.
Sources:
http://chemistry.about.com/od/demonstrationsexperiments/ss/bounceball.htm
http://www.hometrainingtools.com/a/make-a-colorful-bouncy-ball
http://www.pbs.org/parents/crafts-for-kids/super-bouncy-balls/
Materials:
- cup or bowl
- stirrer
- 1 T. white glue
- 1/2 tsp. borax powder
- food coloring
- 1 T. cornstarch
- 2 T. warm water
- In one cup, combine the warm water, cornstarch and borax. In another cup or bowl, put the glue.
- Next, add several drops of food coloring to the white glue and stir well.
- Give your water/borax/cornstarch mixture a good stir to combine all the ingredients (the cornstarch has a tendency to settle on the bottom). Then pour the water mixture into the colored glue.
- Stir and you'll see it immediately start to clump together. Stir a bit more until you have one big, slimy glob.
- Take the glob out of the liquid and begin rolling it between the palms of your hands to form a ball. It'll be sticky at first, so keep a paper towel handy to wipe off your hands occasionally. Soon a rubbery ball will form. (We found that just using our palms seemed to work better than using our whole hands and fingers.)
- Once the stickiness is gone and you have a nice smooth ball, that's it! You can bounce away!
- Borax and glue are a potential health hazard and should not be ingested or sniffed
Everything should be washable after the experiment.
Reflection:
Looking Back:
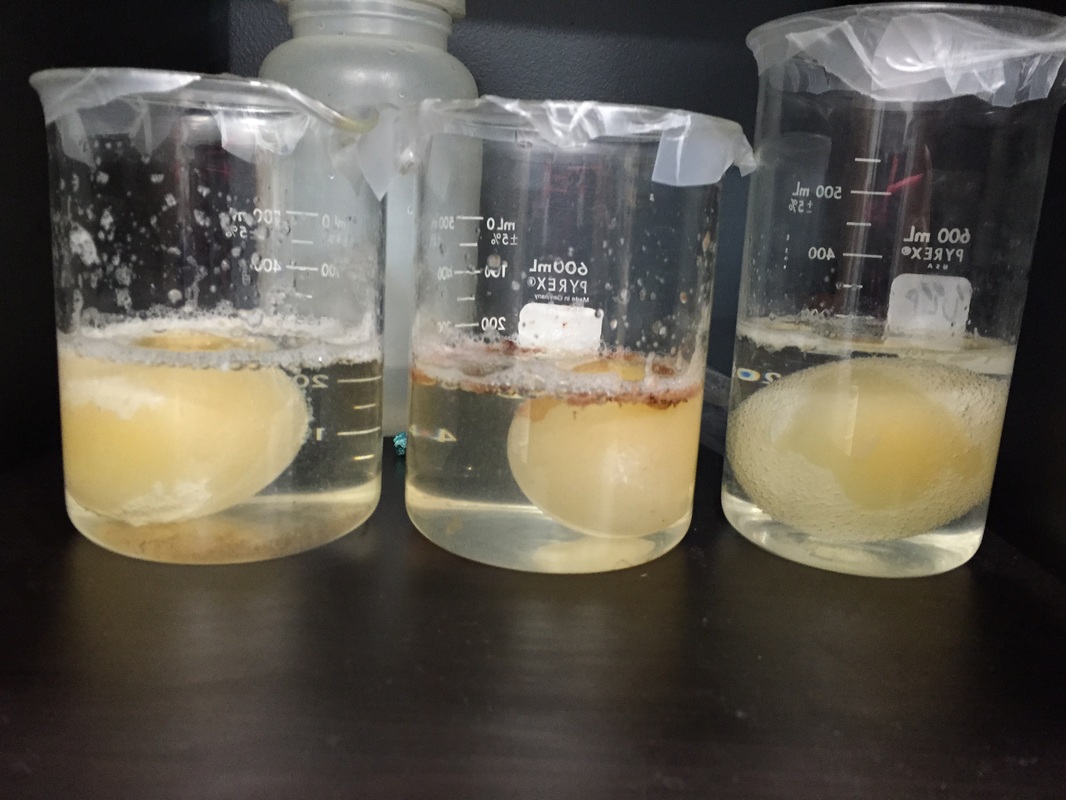
When Daisy and I were trying out this experiment, one of us tried it with 1 table spoon of cornstarch and 2 tablespoons of warm water while the other tried it with 3 table spoons of cornstarch and 4 table spoons of warm water. The ball with 1 tablespoon of cornstarch worked better while the other with 3 tablespoons of cornstarch didn't even bounce at all. Both of the balls felt rubbery and kind of like putty. The ball that didn't bounce didn't even want to form into a ball shape and it seemed as though the glue didn't want to mix with the cornstarch. The ball that bounced, bounced a couple feet high but one time when we were playing with it, I thing we threw it on the floor too hard that the ball didn't bounce but it split in half so it probably got to much pressure.
Looking Beyond:
First what is did for the experiment was combining the corn starch and borax with the water and that made a oobleck type sustance. From my previous lab with the oobleck, I know that the molecules in oobleck changes from a liquid state to a solid state depending on how much pressure id applied to it. After I did that I poured the glue into a separate container and mixed it with food coloring so my ball would be colorful. Then I poured the oobleck stuff into the glue and started stirring. After a few seconds of stirring I've noticed that the liquids have become a solid. This is when I believe that the chemical reaction between the borax and the glue. The molecules of both substances cross linked to become the rubbery ball. After I saw that it became a rubbery substance, I put it in my hand and started forming it's round shape. It took a couple of minutes for the ball to become totally dry and not sticky. Then we started playing with the ball and we noticed that it could bounce up to a couple feet high. Since we made multiply balls, I noticed that different proportions of the materials give different outcomes. More glue would result to a slimier ball, less borax would result to a goopier ball, and more cornstarch would result to a more stretchy and bendy ball.
Looking Inward:
The ball I made was the results that I wanted but the ball that Daisy made was a fail. I think the fact that we used different proportions of cornstarch and water made a difference of how the balls turned out. We both did the same procedure to make the balls and the turn outs were different. I think I performed pretty well in this lab, I followed the instructions carefully and Daisy and I saw how each proportion can change how the ball would turn out. One thing I thought about that made Daisy's ball not bounce was that the glue that we used. The glue I used was elmer's and since we ran out of it, for Daisy's we had to use roseart. I believe that since they are different brands of glue, they contain different ingredients in them too. I remember that a website I went on had emphasis of elmer's glue and said that it worked best.
Looking Outward:
I worked with Daisy, Carmina, and Erika in this lab. I feel we work well together and we have worked together before and most of the time the outcome of those works were great. We communicated and collaborated well to organised the lab and decide which days were good to start the lab. We had to do the lab on two separate days because the day we originally planned the lab, Carmina for got to bring the glue but I brought some but I only had enough for about two balls. So Daisy and I used my glue on that day and Erika was busy with other labs and Carmina had to leave for a track meet. The next day Carmina brought the glue and Daisy got to redo the lab since her first ball didn't work out and Carmina and Erika got to do the lab themselves. I think our group performed okay because all of us kind of did the lab on different days and some of us weren't present when the other was doing the lab. But our group compared our outcomes with each other.
Looking Forward:
If I were to do this lab again I would only use the proportions I used when Daisy and I did the lab because that recipe worked better. Also, I would make sure that the glue we use will be elmer's glue because that gave us a better outcome. Although we got some balls that didn't turn out so well, I liked that we tested different proportions of the recipe and different types of glue because that is what got us thinking of what went wrong. If I were to do this lab again I wouldn't mind working with this group again. I saw that we worked well together but I just want to point out that some of us were lost about the science that was going on so next time when I work with them I want to make sure they did their share of the lab and learn the science behind it instead of just googling afterwards or asking the people in our group that actually researched about it for the lab. I don't really see any way to continue this lab but I know that the concept of polymers connects to many other science labs like slime and putty and I would like to do those labs too.
Takeaways:
For this lab I took away that I have to always remind the person who is bringing the supplies to bring them in early or to put it in their backpack the night before so they can remember. I also took away that I have to make sure that everyone in the group reads through the proposal and learned what exactly they are doing in the lab. I also gained knowledge about polymers and how different materials connect that was new information to me. Also, for the experiment, we forgot to bring teaspoon and table spoon measures so we had to convert to milliliters because that was what we had in the classroom and I took away that if we don't have the correct measuring device ready, we can always covert so we can use the measuring devices available.
When Daisy and I were trying out this experiment, one of us tried it with 1 table spoon of cornstarch and 2 tablespoons of warm water while the other tried it with 3 table spoons of cornstarch and 4 table spoons of warm water. The ball with 1 tablespoon of cornstarch worked better while the other with 3 tablespoons of cornstarch didn't even bounce at all. Both of the balls felt rubbery and kind of like putty. The ball that didn't bounce didn't even want to form into a ball shape and it seemed as though the glue didn't want to mix with the cornstarch. The ball that bounced, bounced a couple feet high but one time when we were playing with it, I thing we threw it on the floor too hard that the ball didn't bounce but it split in half so it probably got to much pressure.
Looking Beyond:
First what is did for the experiment was combining the corn starch and borax with the water and that made a oobleck type sustance. From my previous lab with the oobleck, I know that the molecules in oobleck changes from a liquid state to a solid state depending on how much pressure id applied to it. After I did that I poured the glue into a separate container and mixed it with food coloring so my ball would be colorful. Then I poured the oobleck stuff into the glue and started stirring. After a few seconds of stirring I've noticed that the liquids have become a solid. This is when I believe that the chemical reaction between the borax and the glue. The molecules of both substances cross linked to become the rubbery ball. After I saw that it became a rubbery substance, I put it in my hand and started forming it's round shape. It took a couple of minutes for the ball to become totally dry and not sticky. Then we started playing with the ball and we noticed that it could bounce up to a couple feet high. Since we made multiply balls, I noticed that different proportions of the materials give different outcomes. More glue would result to a slimier ball, less borax would result to a goopier ball, and more cornstarch would result to a more stretchy and bendy ball.
Looking Inward:
The ball I made was the results that I wanted but the ball that Daisy made was a fail. I think the fact that we used different proportions of cornstarch and water made a difference of how the balls turned out. We both did the same procedure to make the balls and the turn outs were different. I think I performed pretty well in this lab, I followed the instructions carefully and Daisy and I saw how each proportion can change how the ball would turn out. One thing I thought about that made Daisy's ball not bounce was that the glue that we used. The glue I used was elmer's and since we ran out of it, for Daisy's we had to use roseart. I believe that since they are different brands of glue, they contain different ingredients in them too. I remember that a website I went on had emphasis of elmer's glue and said that it worked best.
Looking Outward:
I worked with Daisy, Carmina, and Erika in this lab. I feel we work well together and we have worked together before and most of the time the outcome of those works were great. We communicated and collaborated well to organised the lab and decide which days were good to start the lab. We had to do the lab on two separate days because the day we originally planned the lab, Carmina for got to bring the glue but I brought some but I only had enough for about two balls. So Daisy and I used my glue on that day and Erika was busy with other labs and Carmina had to leave for a track meet. The next day Carmina brought the glue and Daisy got to redo the lab since her first ball didn't work out and Carmina and Erika got to do the lab themselves. I think our group performed okay because all of us kind of did the lab on different days and some of us weren't present when the other was doing the lab. But our group compared our outcomes with each other.
Looking Forward:
If I were to do this lab again I would only use the proportions I used when Daisy and I did the lab because that recipe worked better. Also, I would make sure that the glue we use will be elmer's glue because that gave us a better outcome. Although we got some balls that didn't turn out so well, I liked that we tested different proportions of the recipe and different types of glue because that is what got us thinking of what went wrong. If I were to do this lab again I wouldn't mind working with this group again. I saw that we worked well together but I just want to point out that some of us were lost about the science that was going on so next time when I work with them I want to make sure they did their share of the lab and learn the science behind it instead of just googling afterwards or asking the people in our group that actually researched about it for the lab. I don't really see any way to continue this lab but I know that the concept of polymers connects to many other science labs like slime and putty and I would like to do those labs too.
Takeaways:
For this lab I took away that I have to always remind the person who is bringing the supplies to bring them in early or to put it in their backpack the night before so they can remember. I also took away that I have to make sure that everyone in the group reads through the proposal and learned what exactly they are doing in the lab. I also gained knowledge about polymers and how different materials connect that was new information to me. Also, for the experiment, we forgot to bring teaspoon and table spoon measures so we had to convert to milliliters because that was what we had in the classroom and I took away that if we don't have the correct measuring device ready, we can always covert so we can use the measuring devices available.











 RSS Feed
RSS Feed
